Explanation of Electronic Configuration of Elements
Explanation of Electronic Configuration of Elements. The term “electronic configuration” comes to mind if you are curious about the atomic structure of an element. This is a core concept in chemistry and it is crucial for understanding the elements that make up our planet.
Table of Contents
Introduction:
Molecular and atomic structure and behavior are largely explained by electronic configuration as a concept in chemistry.
By using it, one can predict the properties of elements and compounds by describing electrons are arranged in an atom or molecule.
Insight into matter’s properties can be gained by understanding the electronic configurations of elements and compounds.
What is Electronic Configuration?
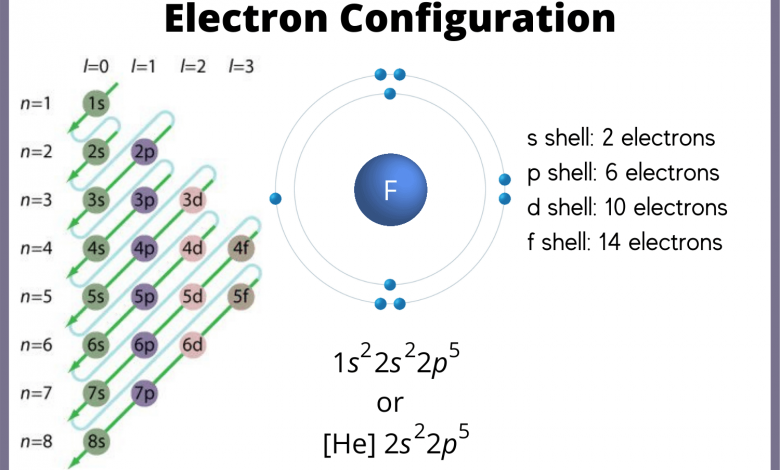
Electron configuration refers to electrons arranged within the atomic orbitals of atoms or ions. It is generally expressed by a set of quantum numbers or notations such as 1s2 2s2 2p6 3s2 3p6 4s2 3d7 for the element chromium (Cr).
Atoms’ chemical and physical properties are determined by their electronic configuration.
Explanation of Electronic Configuration:
The concept of electronic configuration is fundamental to chemistry, which describes how electrons are arranged in molecules and atoms. It is based on quantum mechanics principles and is key to understanding chemical properties.
Arrangement of Electron:
The electronic configuration is the arrangement of electrons around an atom or molecule at its most basic level.
Each electron is contained in a shell or orbital, and the number of electrons in each shell is determined by the element’s atomic number, for example, carbon has an atomic number of 6, so its electronic configuration is 2.
In other words, the first shell contains two electrons, while the second shell contains four electrons.
Atoms contain electrons most likely to interact with other atoms in their outermost shell known as the valence shell.
Electron Located:
Those electrons that are located in the valence shell are called valence electrons. These electrons are those that are involved in chemical reactions. The quantity of valence electrons in an atom or molecule determines its reactivity.
Elements belonging to the same group of the periodic table are generally expected to have similar electronic configurations. These elements will also have the same number of shells and valence electrons.
Consequently, elements in the same group of the periodic table will have the same number of valence electrons since the number of electrons in each shell is determined by the atomic number of each element.
Electronic Configuration Affects:
A molecule or atom’s electronic configuration also affects its chemical properties. Since valence electrons are involved in chemical reactions, the arrangement of these electrons can determine the types of reactions that can take place.
A molecule with strong covalent bonds will be more reactive than one with weak covalent bonds, for example, if the valence electrons are arranged so that they form strong covalent bonds.
The electronic configuration of molecules or atoms is also crucial to understanding chemical reactivity. Different atoms and molecules may have different chemical reactivity depending on their electronic configuration.
As a result of the arrangement of the valence electrons in the molecule or atom, some atoms or molecules may be more reactive than others within a group of the periodic table.
How Electronic Configuration of Elements is Organized?
Electronic configurations of elements are organized into a system, the periodic table, in which elements are arranged based on their atomic numbers into rows and columns.
Elements whose properties are similar are grouped in columns, whereas elements whose properties differ are grouped in rows. As an example, elements with atomic number 1 are grouped in the first column of the periodic table.
Chemists utilize the electronic configuration of elements to predict their chemical and physical properties This facilitates the development of new compounds and the prediction of their characteristics.
Examples of Electronic Configuration:
A hydrogen atom’s ground state configuration is illustrated by the notation 1s1 for the configuration with one electron in the 1s orbital. This configuration is an example of an electronic configuration. As part of the notation of an atom’s electronic configuration, the Aufbau Principle is used, which explains how the lowest energy orbitals are filled first.
Use of Orbital Diagram:
A diagram of the orbital configuration of elements can also be used to illustrate their electronic configuration. An orbital diagram is constructed from circles that represent orbitals, with the innermost circles representing the lowest energy orbitals.
A hydrogen atom’s ground state configuration can be represented as follows based on the number of arrows in each orbital. The orbital diagram below illustrates the electron’s position in the 1s orbital of the hydrogen atom.
A similar orbital diagram can also be used to represent the electronic configurations of more complex atoms. As an example, carbon has a configuration of 1s2 2s2 2p2.
This notation indicates that one electron occupies the 1s orbital, two electrons occupy the 2s orbital, and two electrons occupy the 2p orbital.
The Electronic Configuration can be represented as:
The electronic configuration of an atom can also be represented using noble gas notation. The noble gas symbol used in this notation is that of the noble gas that is in the period preceding the atom in question.
For example, oxygen’s electronic configuration is [He] 2s2. According to the formula, the oxygen atom consists of four electrons in the 2p orbital and two electrons in the 2s orbital.
Conclusion:
Atoms and molecules are composed of atoms and molecules that are configured electronically.
To correctly classify elements, predict their properties, and understand how they interact with other elements and molecules, it is necessary to understand the arrangement of electrons around the nucleus of an atom.




